Ensuring the quality and authenticity of Anabolic Androgenic Steroids (AAS) products is paramount for Euro-Pharmacies to uphold safety standards and maintain consumer trust. In the competitive pharmaceutical industry, adherence to stringent regulations and implementation of robust quality assurance measures are essential for the production and distribution of Anabolic Androgenic Steroid products. This article explores the various strategies and practices that Euro-Pharmacies can employ to ensure the quality and authenticity of their AAS products, ranging from compliance with regulatory frameworks to the implementation of good manufacturing practices and quality control measures.
By prioritizing quality assurance, Euro-Pharmacies can not only enhance product efficacy and safety but also mitigate the risks associated with counterfeit and adulterated products in the market.
1. Importance of Quality Assurance in Euro-Pharmacies
Understanding the Risks of Poor Quality in AAS Products
When it comes to anabolic androgenic steroids (AAS), the stakes are high for Euro-Pharmacies to prioritize quality assurance. Poor quality AAS products can not only be ineffective but also pose serious health risks to consumers. Ensuring the purity and authenticity of these products is crucial for the safety and well-being of users.
Building Trust and Reputation through Quality Assurance
Quality assurance is not just about meeting regulatory standards; it’s about earning the trust of consumers. Euro-Pharmacies that consistently deliver high-quality AAS products build a strong reputation in the market. Trust is a currency that is hard to earn but easy to lose, making quality assurance a non-negotiable aspect for Euro-Pharmacies.
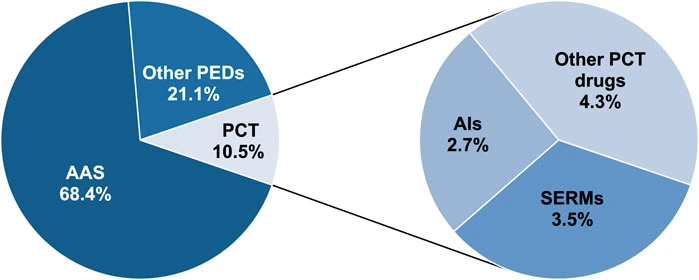
2. Regulatory Framework for Anabolic Androgenic Steroid Products in Europe and Worldwide
Evaluating EU Regulations on Anabolic Androgenic Steroids
In Europe, the regulatory landscape for AAS products is stringent. Euro-Pharmacies must navigate complex regulations governing the production, distribution, and marketing of AAS. Understanding and adhering to these regulations is crucial for operating legally and ethically in the market.
Compliance with Testing and Reporting Requirements
Pharmacovigilance is a key component of ensuring the safety of Anabolic Androgenic Steroid products. Euro-Pharmacies must comply with reporting requirements for adverse effects and monitor the quality and safety of their products throughout their lifecycle. By being vigilant and proactive, Euro-Pharmacies can contribute to maintaining a safe supply chain of AAS products worldwide.
3. Implementing Good Manufacturing Practices (GMP) in Euro-Pharmacies
Key Principles of GMP for AAS Production
Good Manufacturing Practices (GMPs) are the foundation of quality assurance in pharmaceutical manufacturing. Euro-Pharmacies must adhere to strict GMP guidelines to ensure the consistency, quality, and safety of their AAS products. By implementing GMP principles, Euro-Pharmacies can streamline their production process and reduce the risk of contamination or errors.
Training and Personnel Qualifications
Quality assurance starts with the people behind the products. Euro-Pharmacies need to invest in training and developing their personnel to ensure they have the knowledge and skills to maintain GMP standards. Well-trained employees are the frontline defense against quality issues and play a crucial role in upholding the integrity of Anabolic Androgenic Steroid production.
4. Quality Control Measures for Anabolic Androgenic Steroid Products
Testing Procedures and Specifications
Quality control is about more than just checking boxes; it’s about rigorous testing and adherence to specifications. Euro-Pharmacies must have robust testing procedures in place to verify the identity, purity, and potency of their Anabolic Androgenic Steroid products. By testing at various stages of production, Euro-Pharmacies can catch any deviations early and ensure consistent quality.
In the fast-paced world of Euro-Pharmacies, where quality and authenticity are paramount, a comprehensive approach to quality assurance is not just a best practice but a necessity. By understanding and implementing stringent quality control measures, Euro-Pharmacies can safeguard their reputation, protect consumers, and contribute to a safer and more regulated market for Anabolic Androgenic Steroid products in Europe 5. Ensuring Authenticity through Traceability and Documentation:
TEST RESULTS that may be verified by 3rd-party testing
When it comes to Euro-Pharmacies and their AAS products, ensuring authenticity is key. One way to achieve this is through robust track and trace systems. By implementing these systems, Euro-Pharmacies can keep a close eye on their products throughout the supply chain, from production to distribution. This not only helps in verifying the authenticity of the products but also in quickly identifying and addressing any issues that may arise.
Moreover, documenting the integrity of the entire supply chain is crucial. By maintaining detailed records of every step in the journey of their AAS products, Euro-Pharmacies can establish a transparent and traceable process. This documentation not only aids in quality control but also builds trust with customers and regulatory bodies.
6. Role of Quality Testing and Certification in Euro-Pharmacies
Quality testing and certification play a significant role in upholding the standards of Euro-Pharmacies’ AAS products. Third-party testing adds an extra layer of assurance regarding the quality and authenticity of the products. Euro-Pharmacies can showcase their commitment to meeting industry standards and regulations that AAS users worldwide need and expect..
Utilizing analytical techniques for quality control is also essential. By employing advanced testing methods, Euro-Pharmacies can detect any impurities or deviations in their products, ensuring that only quality Anabolic Androgenic Steroid products reach the market. This proactive approach not only safeguards the reputation of Euro-Pharmacies but also protects consumer health.
7. Strategies for Preventing Counterfeiting and Adulteration
Counterfeiting and adulteration pose significant risks to Euro-Pharmacies and their Anabolic Androgenic Steroid products. To combat these threats, securing packaging and labels is paramount. By implementing tamper-evident packaging and incorporating security features in labels, Euro-Pharmacies can deter counterfeiters and protect the authenticity of their products.
In addition, leveraging anti-counterfeiting technologies can be a game-changer. From holograms to QR codes, these technologies make it easier for consumers and authorities to verify the legitimacy of AAS products. By staying one step ahead of counterfeiters, Euro-Pharmacies can maintain the integrity of their brand and products.
8. Continuous Improvement and Compliance in Euro-Pharmacies
Continuous improvement and compliance are pillars of success for Euro-Pharmacies. Utilizing feedback mechanisms and quality metrics allows Euro-Pharmacies to gather valuable insights from customers. By listening to feedback and analyzing performance metrics, Euro-Pharmacies can identify areas for improvement and implement necessary changes to enhance product quality.
Moreover, adapting to regulatory changes and industry trends is crucial. Staying informed about new regulations and emerging trends in the AAS market enables Euro-Pharmacies to remain compliant and competitive. By demonstrating agility and a willingness to evolve, Euro-Pharmacies can stay ahead of the curve and continue delivering high-quality Anabolic Androgenic Steroid products to their customers.
In conclusion, by proactively addressing quality assurance and authenticity concerns in the production of AAS products, Euro-Pharmacies can uphold their commitment to delivering safe and reliable pharmaceuticals to consumers. Continuous improvement, compliance with regulations, and the adoption of best practices in quality control and testing are integral to sustaining the integrity of AAS products in the market. Through these efforts, Euro-Pharmacies can instill confidence in their products, build a strong reputation, and contribute to the overall safety and efficacy of AAS usage within the healthcare landscape.
Why is quality assurance important in Anabolic Androgenic Steroid (AAS) products?
Quality assurance ensures that AAS products are safe, effective, and free from contamination. It protects consumers from counterfeit or adulterated steroids while maintaining the reputation of trusted pharmaceutical companies like Euro-Pharmacies.
How does Euro-Pharmacies verify the authenticity of Anabolic Androgenic Steroid products?
Euro-Pharmacies uses traceability systems, secure packaging, and third-party testing to verify the authenticity of its AAS products. This ensures transparency and consumer trust.
What role do Good Manufacturing Practices (GMP) play in AAS production?
GMP guidelines help Euro-Pharmacies maintain consistency, quality, and safety in Anabolic Androgenic Steroid manufacturing. They reduce the risks of contamination, errors, and poor-quality products.
How can consumers identify genuine Euro-Pharmacies AAS products?
Consumers should look for tamper-evident packaging, security features such as holograms or QR codes, and verify batch numbers against Euro-Pharmacies’ official records.
What steps are taken to prevent counterfeit Anabolic Androgenic Steroid products?
Euro-Pharmacies implements advanced labeling technologies, secure packaging, and track-and-trace systems to prevent counterfeit and adulterated AAS products from entering the market.

